Main article: Bacillus anthracis
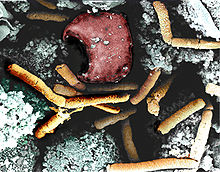
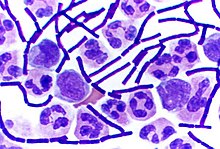
Bacillus anthracis is a rod-shaped, Gram-positive, aerobic bacterium about 1 by 9 μm in size. It was shown to cause disease by Robert Koch in 1876 when he took a blood sample from an infected cow, isolated the bacteria and put them into a mouse.[21] The bacterium normally rests in endospore form in the soil, and can survive for decades in this state. Herbivores are often infected whilst grazing, especially when eating rough, irritant, or spiky vegetation: the vegetation has been hypothesized to cause wounds within thegastrointestinal tract permitting entry of the bacterial endospores into the tissues, though this has not been proven. Once ingested or placed in an open wound, the bacterium begins multiplying inside the animal or human and typically kills the host within a few days or weeks. The endospores germinate at the site of entry into the tissues and then spread by the circulation to the lymphatics, where the bacteria multiply.
The production of two powerful exotoxins and lethal toxin by the bacteria causes death. Veterinarians can often tell a possible anthrax-induced death by its sudden occurrence, and by the dark, nonclotting blood that oozes from the body orifices. Most anthrax bacteria inside the body after death are outcompeted and destroyed by anaerobic bacteria within minutes to hours post mortem. However, anthrax vegetative bacteria that escape the body via oozing blood or through the opening of the carcass may form hardy spores. One spore forms per one vegetative bacterium. The triggers for spore formation are not yet known, though oxygen tension and lack of nutrients may play roles. Once formed, these spores are very hard to eradicate.
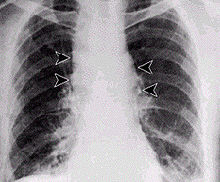
The infection of herbivores (and occasionally humans) by the inhalational route normally proceeds as follows: Once the spores are inhaled, they are transported through the air passages into the tiny air sacs (alveoli) in the lungs. The spores are then picked up by scavenger cells (macrophages) in the lungs and are transported through small vessels (lymphatics) to the lymph nodes in the central chest cavity (mediastinum). Damage caused by the anthrax spores and bacilli to the central chest cavity can cause chest pain and difficulty in breathing. Once in the lymph nodes, the spores germinate into active bacilli that multiply and eventually burst the macrophages, releasing many more bacilli into the bloodstream to be transferred to the entire body. Once in the blood stream, these bacilli release three proteins named lethal factor, edema factor, and protective antigen. The three are not toxic by themselves, but the combination is incredibly lethal to humans.[22] Protective antigen combines with these other two factors to form lethal toxin and edema toxin, respectively. These toxins are the primary agents of tissue destruction, bleeding, and death of the host. If antibiotics are administered too late, even if the antibiotics eradicate the bacteria, some hosts will still die of toxemia because the toxins produced by the bacilli remain in their system at lethal dose levels.
The lethality of the anthrax disease is due to the bacterium's two principal virulence factors: the poly-D-glutamic acid capsule, which protects the bacterium from phagocytosis by host neutrophils, and the tripartite protein toxin, called anthrax toxin. Anthrax toxin is a mixture of three protein components: protective antigen (PA), edema factor (EF), and lethal factor (LF). PA plus LF produces lethal toxin, and PA plus EF produces edema toxin. These toxins cause death and tissue swelling (edema), respectively.
To enter the cells, the edema and lethal factors use another protein produced by B. anthracis called protective antigen, which binds to two surface receptors on the host cell. A cell protease then cleaves PA into two fragments: PA20 and PA63. PA20 dissociates into the extracellular medium, playing no further role in the toxic cycle. PA63 then oligomerizes with six other PA63 fragments forming a heptameric ring-shaped structure named a prepore. Once in this shape, the complex can competitively bind up to three EFs or LFs, forming a resistant complex.[22] Receptor-mediated endocytosis occurs next, providing the newly formed toxic complex access to the interior of the host cell. The acidified environment within the endosome triggers the heptamer to release the LF and/or EF into the cytosol.[23] It is unknown how exactly the complex results in the death of the cell.
Edema factor is a calmodulin-dependent adenylate cyclase. Adenylate cyclase catalyzes the conversion of ATP into cyclic AMP (cAMP) and pyrophosphate. The complexation of adenylate cyclase with calmodulin removes calmodulin from stimulating calcium-triggered signaling, thus inhibiting the immune response.[22] To be specific, LF inactivatesneutrophils (a type of phagocytic cell) by the process just described so they cannot phagocytose bacteria. Throughout history, lethal factor was presumed to caused macrophages to make TNF-alpha and interleukin 1, beta (IL1B). TNF-alpha is a cytokine whose primary role is to regulate immune cells, as well as to induce inflammation andapoptosis or programmed cell death. Interleukin 1, beta is another cytokine that also regulates inflammation and apoptosis. The overproduction of TNF-alpha and IL1B ultimately leads to septic shock and death. However, recent evidence indicates anthrax also targets endothelial cells that line serous cavities such as the pericardial cavity, pleural cavity, and the peritoneal cavity, lymph vessels, and blood vessels, causing vascular leakage of fluid and cells, and ultimately hypovolemic shock and septic shock.
Exposure[edit]
Occupational exposure to infected animals or their products (such as skin, wool, and meat) is the usual pathway of exposure for humans. Workers who are exposed to dead animals and animal products are at the highest risk, especially in countries where anthrax is more common. Anthrax in livestock grazing on open range where they mix with wild animals still occasionally occurs in the United States and elsewhere. Many workers who deal with wool and animal hides are routinely exposed to low levels of anthrax spores, but most exposure levels are not sufficient to develop anthrax infections. The body's natural defenses presumably can destroy low levels of exposure. These people usually contract cutaneous anthrax if they catch anything. Throughout history, the most dangerous form of inhalational anthrax was called woolsorters' disease because it was an occupational hazard for people who sorted wool. Today, this form of infection is extremely rare, as almost no infected animals remain. The last fatal case of natural inhalational anthrax in the United States occurred in California in 1976, when a home weaver died after working with infected wool imported from Pakistan. To minimize the chance of spreading the disease, the deceased was transported to UCLA in a sealed plastic body bag within a sealed metal container for autopsy.[24]
In November 2008, a drum maker in the United Kingdom who worked with untreated animal skins died from anthrax.[25] Gastrointestinal anthrax is exceedingly rare in the United States, with only one case on record, reported in 1942, according to the Centers for Disease Control and Prevention.[17] In December 2009, an outbreak of anthrax occurred amongst heroin addicts in Glasgow, Scotland, resulting in 14 deaths.[26] The source of the anthrax is believed to be dilution of the heroin with bone meal in Afghanistan.[27]
Also during December 2009, the New Hampshire Department of Health and Human Services confirmed a case of gastrointestinal anthrax in an adult female. The CDCinvestigated the source and the possibility that it was contracted from an African drum recently used by the woman taking part in a drumming circle.[28] The woman apparently inhaled anthrax [in spore form] from the hide of the drum. She became critically ill, but with gastrointestinal anthrax rather than inhaled anthrax, which made her unique in American medical history. The building where the infection took place was cleaned and reopened to the public and the woman recovered. Jodie Dionne-Odom, New Hampshire state epidemiologist, stated, "It is a mystery. We really don't know why it happened."[29]
Mode of infection[edit]
Anthrax can enter the human body through the intestines (ingestion), lungs (inhalation), or skin (cutaneous) and causes distinct clinical symptoms based on its site of entry. In general, an infected human will be quarantined. However, anthrax does not usually spread from an infected human to a noninfected human. But, if the disease is fatal to the person's body, its mass of anthrax bacilli becomes a potential source of infection to others and special precautions should be used to prevent further contamination. Inhalational anthrax, if left untreated until obvious symptoms occur, may be fatal.
Anthrax can be contracted in laboratory accidents or by handling infected animals or their wool or hides. It has also been used in biological warfare agents and by terrorists to intentionally infect as exemplified by the 2001 anthrax attacks.
No comments:
Post a Comment